Metal- and Solvent-Free Synthesis of Tris(indolyl)methanes via the Reaction of Indoles with Trimethyl Orthoacetate
Keywords:
tris(indolyl)methane, trimethyl orthoacetate, 𝘱-toluenesulfonic acid, Friedel–Crafts-type electrophilic substitution, solvent-free synthesisAbstract
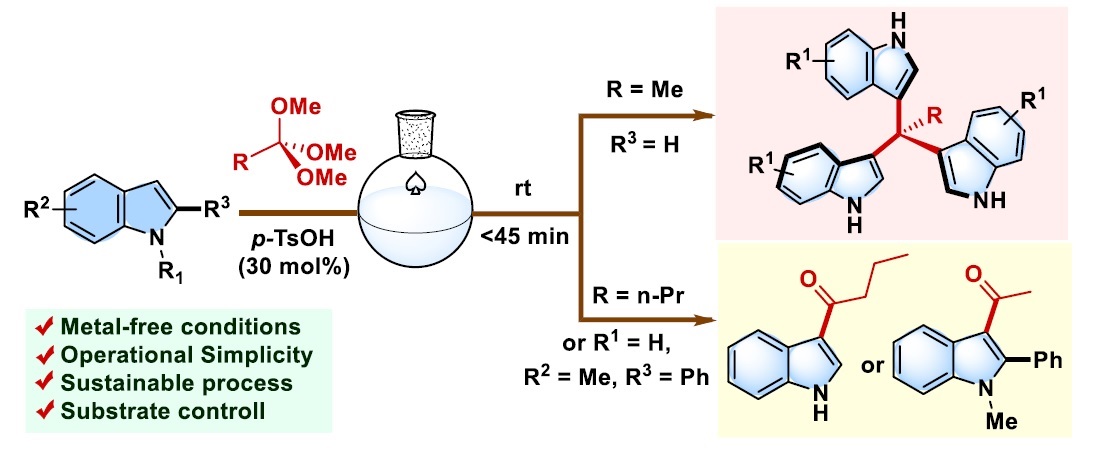
Background and Objectives: Tris(indolyl)methanes (TIMs) are important heterocyclic scaffolds widely found in biologically active molecules and functional organic materials. Indole-containing compounds are prevalent in natural products, pharmaceuticals, and agrochemicals, and exhibit diverse biological activities, including anticancer, antimicrobial, antiviral, and anti-inflammatory effects. In particular, TIM derivatives have attracted considerable attention due to their pharmacological potential and their utility as key intermediates in organic synthesis, as well as their applications in materials science such as organic electronics and molecular sensors. TIMs are typically synthesized via acid-catalyzed Friedel–Crafts reactions between indoles and electrophilic carbon sources such as orthoesters. However, many reported methods rely on strong mineral acids, expensive metal catalysts, elevated temperatures, or large amounts of organic solvents, which pose environmental concerns and operational limitations. Although recent advances in green chemistry have promoted solvent-free and metal-free approaches, the development of simple, efficient, and environmentally benign methods for TIM synthesis remains a significant challenge. Therefore, this study aims to develop a straightforward and efficient method for the synthesis of tris(indolyl)methanes via the reaction of indoles with trimethyl orthoacetate under solvent-free and metal-free conditions, providing a practical and sustainable alternative with operational simplicity and improved environmental compatibility.
Methodology: The synthesis of tris(indolyl)methane derivatives was achieved through a Friedel–Crafts-type electrophilic substitution reaction between indoles and trimethyl orthoacetate. In this study, p-toluenesulfonic acid (p-TSA) was employed as an inexpensive and readily available organic acid catalyst to promote the reaction. The reactions were carried out under solvent-free conditions at room temperature in order to enhance the environmental compatibility of the process and to simplify the experimental procedure. Typically, indole (1 mmol) and trimethyl orthoacetate (1 mmol) were mixed in the presence of 30 mol% p-toluenesulfonic acid, and the reaction progress was monitored over time. To evaluate the generality and applicability of the developed protocol, a variety of indole substrates bearing different substituents on the aromatic ring were examined. The corresponding tris(indolyl)methane products were isolated and characterized using spectroscopic techniques.
Main Results: The developed protocol successfully afforded the desired tris(indolyl)methane derivatives within relatively short reaction times ranging from 30 minutes to 2 hours. The products were obtained in moderate to good yields, typically ranging from 40% to 80%, depending on the electronic nature of the substituents on the indole ring. Electron-donating and electron-withdrawing substituents were generally tolerated under the optimized conditions, demonstrating the versatility of the reaction. Importantly, the transformation proceeded efficiently under mild conditions without the need for metal catalysts, external solvents, or elevated temperatures. The solvent-free reaction environment also contributed to a simpler reaction setup and reduced waste generation. These results indicate that trimethyl orthoacetate serves as an effective electrophilic partner for the formation of tris(indolyl)methane derivatives through a Friedel–Crafts-type process.
Conclusions : This study describes a convenient and environmentally friendly method for the synthesis of tris(indolyl)methane derivatives through the reaction of indoles with trimethyl orthoacetate. The protocol employs p-toluenesulfonic acid as an inexpensive catalyst and operates efficiently under metal-free and solvent-free conditions at room temperature. The method provides moderate to good yields and exhibits a reasonable substrate scope with various substituted indoles. Compared with previously reported procedures, the present approach offers several advantages, including operational simplicity, mild reaction conditions, reduced use of hazardous reagents, and improved environmental compatibility. Therefore, this method represents a practical and sustainable alternative for the preparation of tris(indolyl)methane derivatives and may be useful for future applications in medicinal chemistry and organic synthesis.
References
Akgün, E., Tunali, M., & Pindur, U. (1987). Proton acid-catalysed acylation of indoles by 2-alkoxy-1, 3 dioxolanes. Archievs Pharmacy, 320, 397–401.
Akgün, H., & Hofmann, A. W. (1983). Reaktionen von elektronenreichen Heterocyclen mit Orthocarbonsäure-Derivaten. I. Zum Reaktionsverhalten von acyclischen Orthoestern mit Indolen unter Säurekatalyse. Journal of Heterocyclic Chemistry, 20, 1135–1140.
El‐Sayed, M. T., Mahmoud, K., Hilgeroth, A., & Fakhr, I. M. (2016). Synthesis of novel indolo‐spirocyclic compounds. Journal of Heterocyclic Chemistry, 53(1), 188–196.
Garbe, T. R., Kobayashi, M., Shimizu, N., Takesue, N., Ozawa, M., & Yukawa, H. (2000). Indolyl carboxylic acids by condensation of indoles with α-keto acids. Journal of Natural Products, 63(5), 596–598.
Charoensawas, K., Yimyaem, J., Sirion, U., Suksai, C., Keawin, T., & Jaratjaroonphong, J. (2025). Boron-catalyzed formylation of indoles using trimethyl orthoformate as carbonyl source. ACS Omega, 10(20), 20497–20508.
Khaksar, S., Vahdat, S. M., Gholizadeh, M., & Talesh, S. M. (2012). An expeditious and efficient synthesis of symmetrical tris (indolyl) methanes under catalyst-free conditions in fluorinated alcohols. Journal of Fluorine Chemistry, 136, 8–11.
Lavrenov, S. N., Luzikov, Y. N., Bykov, E. E., Reznikova, M. I., Stepanova, E. V., Glazunova, V. A., Volodina, Y. L., Tatarsky Jr, V. V., Shtil, A. A., & Preobrazhenskaya, M. N. (2010). Synthesis and cytotoxic potency of novel tris(1-alkylindol-3-yl)methylium salts: Role of N-alkyl substituents. Bioorganic & Medicinal Chemistry, 18(18), 6905–6913.
Lavrenov, S. N., Isakova, E. B., Panov, A. A., Simonov, A. Y., Tatarskiy, V. V., & Trenin, A. S. (2020).
N-(Hydroxyalkyl) Derivatives of tris (1 H-indol-3-yl) methylium Salts as Promising Antibacterial Agents: Synthesis and Biological Evaluation. Pharmaceuticals, 13(12), 469.
Ramkissoon, A., Seepersaud, M., Maxwell, A., Jayaraman, J., & Ramsubhag, A. (2020). Isolation and antibacterial activity of indole alkaloids from Pseudomonas aeruginosa UWI-1. Molecules, 25(16), 3744.
Shiri, M., Zolfigol, M. A., Kruger, H. G., & Tanbakouchian, Z. (2010). Bis-and trisindolylmethanes (BIMs and TIMs). Chemical reviews, 110(4), 2250–2293.
Trenin, A. S., Isakova, E. B., Treshchalin, M. I., Polozkova, V. A., Mirchink, E. P., Panov, A. Y., Bychkova, O. P., Tatarskiy, V. V., & Lavrenov, S. N. (2022). Evaluation of New Antimicrobial Agents Based on tris (1 H-Indol-3-yl) methylium Salts: Activity, Toxicity, Suppression of Experimental Sepsis in Mice. Pharmaceuticals, 15(2), 118.
Tuengpanya, S., Chantana, C., Sirion, U., Siritanyong, W., Srisook, K., & Jaratjaroonphong, J. (2018). One-pot solvent-free synthesis of triaryl- and triheteroarylmethanes by Bi(OTf)3-catalyzed Friedel-Crafts reaction of arenes/ heteroarenes with trialkyl orthoformates. Tetranhedron, 74, 4373–4380.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Faculty of Science, Burapha University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Burapha Science Journal is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0) licence, unless otherwise stated. Please read our Policies page for more information




