Hydrothermal Synthesis of Nitrogen-Doped Carbon Dots from Mangosteen Peel: Structural, Optical and Antioxidant Properties
Keywords:
nitrogen doped carbon dots, nitantioxidant activity, urea, hydrothermalAbstract
Background and Objectives: Carbon dots are nanoscale materials primarily composed of carbon atoms. They can be easily synthesized using carbon-rich precursors. In recent years, natural materials have increasingly been used as precursors for the synthesis of carbon dots due to their abundance, low cost, and environmental friendliness. The eastern region of Thailand is one of the major mangosteen cultivation areas in the country. Each year, a large amount of mangosteen peel waste is generated from consumption and food processing activities. Therefore, this research aims to add value to this agricultural waste by utilizing mangosteen peel as a carbon source for the synthesis of nitrogen-doped carbon dots. In this study, nitrogen-doped carbon dots with different nitrogen ratios (MCN1 and MCN2) were synthesized from mangosteen peel using a hydrothermal method, with urea as the nitrogen precursor. The structural characteristics, optical properties, and antioxidant activities of the synthesized nanoparticles were systematically investigated and compared.
Methodology: Nitrogen-doped carbon dots derived from mangosteen peel with different nitrogen ratios (MCN1 and MCN2) were synthesized using a hydrothermal method, with urea employed as the nitrogen source. The characterization of both nanoparticles was carried out using various analytical techniques for example ATR–FTIR , Raman spectrometry, SEM–EDX, HR-TEM, zeta potential, UV–visible spectrophotometry and fluorescence spectrophotometry. The antioxidant activities of MCN1 and MCN2 nanoparticles were evaluated using several standard assays, including the DPPH radical scavenging assay, the ABTS radical cation decolorization assay, the ferric reducing antioxidant power (FRAP) assay, and total phenolic content analysis.
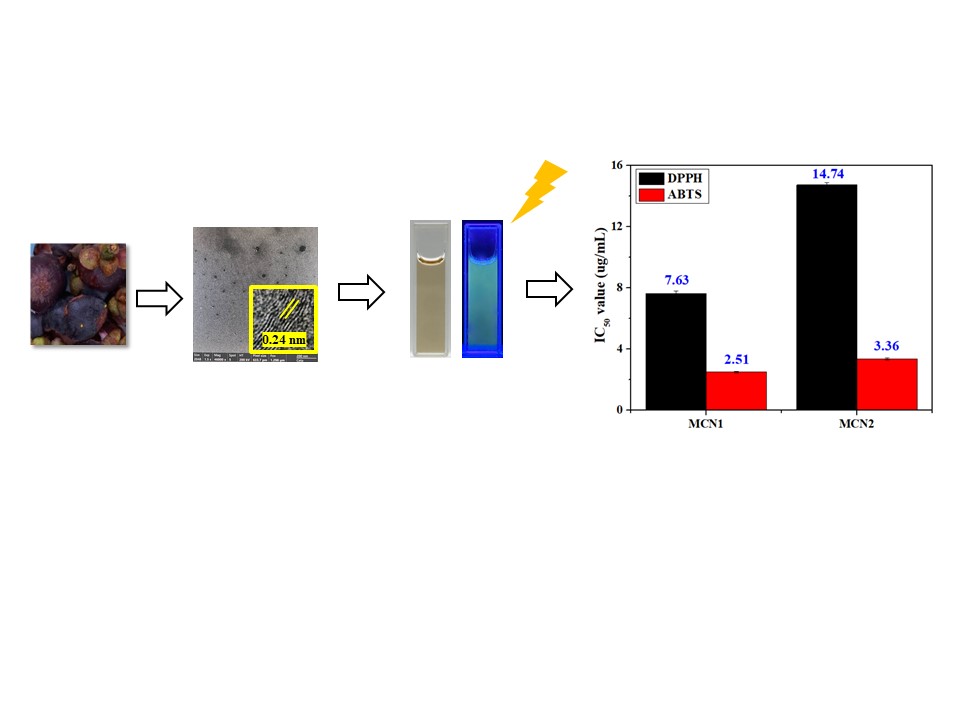
Main Results: In this study, carbon dot particles were synthesized from mangosteen peel via a hydrothermal method, with nitrogen atoms doped at different ratios (MCN1 and MCN2). The objective was to investigate the effect of nitrogen content on the properties of the resulting carbon dots. Structural characteristics and physicochemical properties of the prepared carbon dots were examined using spectroscopic techniques and related structural characterization methods. The results revealed that increasing the nitrogen content led to a higher number of defect sites within the carbon structure. In addition, a higher nitrogen concentration influenced the surface charge properties of the particles, resulting in a less negative surface charge compared with particles containing lower nitrogen content. This alteration in surface charge directly affected the antioxidant activity. The antioxidant evaluation demonstrated that MCN1 particles, which contained a lower nitrogen content than MCN2, exhibited significantly higher antioxidant activity. These findings indicate that the structural features of carbon dots and the nitrogen composition play a crucial role in determining their antioxidant performance.
Conclusions: In this study, nitrogen-doped carbon dot particles were successfully synthesized from mangosteen peel via a hydrothermal method using different nitrogen doping ratios (MCN1 and MCN2). Structural characterization of the synthesized carbon dots using spectroscopic techniques confirmed that a higher nitrogen content resulted in an increased number of structural defect sites. This structural modification also led to a decrease in the negative surface charge of the particles. The change in surface charge was found to have a direct effect on antioxidant activity. Notably, MCN1 particles, which contained a lower nitrogen content than MCN2, exhibited higher antioxidant activity. Therefore, it can be concluded that MCN1 carbon dots possess properties suitable for applications in inhibiting oxidative reactions. Especially, MCN1 show strong potential for use in food packaging materials that require antioxidant functionality to reduce oxidative reactions during food storage. Such materials could contribute to extending the shelf life of food products, minimizing quality deterioration, and enhancing food safety for consumers. Furthermore, this approach represents a promising strategy for valorizing agricultural waste, such as mangosteen peel, by converting it into high-value functional materials with potential industrial and environmental benefits.
References
Amloy, S., Lukprang, T., Lertworapreecha, M., & Preechaburana, P. (2024). Green synthesis of carbon dots from mangosteen peel for fluorescent cancer cells. Journal of Metals, Materials and Minerals, 34(2), 1-8.
Brand-Williams, W., Cuvelier, M. E., & Berset, C. L. W. T. (1995). Use of a free radical method to evaluate antioxidant activity. LWT-Food Science and Technology, 28(1), 25–30.
Chen, X., Zhang, Z., Wu, J., Wang, J., & Gao, A. (2021). Synthesizing red fluorescent carbon dots from rigid polycyclic conjugated molecules: Dual-mode sensing and bioimaging in biochemical applications. Particle & Particle Systems Characterization, 38(7), 2100076.
Chen, Y., Guo, Y., Xing, Z., Bai, L., Yang, W., Wu, Q., & Zhang, G. (2025). Nitrogen-doped carbon dots derived from mangosteen peel for the detection of Cr2O72- and vanillin. Journal of Photochemistry and Photobiology A: Chemistry, 458, 115945.
Chen, Y. X., Tang, Z., Feng, L. Y., Liang, L. L., Shang, D. H., Li, H. Q., & Chen, S. X. (2025). Highly sensitive and selective dual-signal ratiometric fluorescent sensor based on N,S co-doped carbon dots and rhodamine B for Pb2+ ions detection in industrial wastewater. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 127295.
Das, P., Ganguly, S., Mondal, S., Bose, M., Das, A. K., Banerjee, S., & Das, N. C. (2018). Heteroatom doped photoluminescent carbon dots for sensitive detection of acetone in human fluids. Sensors and Actuators B: Chemical, 266, 583-593.
Das, P., Ganguly, S., Ahmed, S. R., Sherazee, M., Margel, S., Gedanken, A., & Rajabzadeh, A. R. (2022). Carbon dot biopolymer-based flexible functional films for antioxidant and food monitoring applications. ACS Applied Polymer Materials, 4(12), 9323–9340.
Dechsri, K., Pengnam, S., Charoenying, T., Nattapulwat, N., Ngawhirunpat, T., Rojanarata, T., & Opanasopit, P. (2025). Fabrication of nitrogen and sulfur co-doped carbon dots for antioxidant applications. Science, Engineering and Health Studies.
Dong, G., Lang, K., Ouyang, H., Zhang, W., Bai, L., Chen, S., & Zhao, X. (2020). Facile synthesis of N,P-doped carbon dots from maize starch via a solvothermal approach for the highly sensitive detection of Fe3+. RSC Advances, 10(55), 33483–33489.
Fawaz, W., Hasian, J., & Alghoraibi, I. (2023). Synthesis and physicochemical characterization of carbon quantum dots produced from folic acid. Scientific Reports, 13(1), 18641.
Gedda, G., Sankaranarayanan, S. A., Putta, C. L., Gudimella, K. K., Rengan, A. K., & Girma, W. M. (2023). Green synthesis of multifunctional carbon dots from medicinal plant leaves for antimicrobial, antioxidant, and bioimaging applications. Scientific Reports, 13, 6371.
Huang, Z., Li, J., & Li, L. S. (2025). Ethylenediamine-assisted preparation of carbon dots from biomass for highly sensitive detection of levodopa. RSC Advances, 15(1), 420–427.
Jayaweera, S., Yin, K., Hu, X., & Ng, W. J. (2019). Facile preparation of fluorescent carbon dots for label-free detection of Fe3+. Journal of Photochemistry and Photobiology A: Chemistry, 370, 156–163.
Kadian, S., Manik, G., Kalkal, A., Singh, M., & Chauhan, R. P. (2019). Effect of sulfur doping on fluorescence and quantum yield of graphene quantum dots. Nanotechnology, 30(43), 435704.
Khan, A., Ezati, P., & Rhim, J. W. (2023). Chitosan/starch-based active packaging film with N,P-doped carbon dots for meat packaging. ACS Applied Biomaterials, 6(3), 1294–1305.
Konar, S., Kumar, B. P., Mahto, M. K., Samanta, D., Shaik, M. A. S., Shaw, M., & Pathak, A. (2019). N-doped carbon dots as fluorescent probe for detection of cysteamine. Sensors and Actuators B: Chemical, 286, 77–85.
Kukongviriyapan, U., Luangaram, S., Leekhaosoong, K., Kukongviriyapan, V., & Preeprame, S. (2007). Antioxidant and vascular protective activities of Cratoxylum formosum, Syzygium gratum and Limnophila aromatica. Biological and Pharmaceutical Bulletin, 30(4), 661–666.
Mao, X. J., Zheng, H. Z., Long, Y. J., Du, J., Hao, J. Y., Wang, L. L., & Zhou, D. B. (2010). Study on fluorescence characteristics of carbon dots. Spectrochimica Acta Part A, 75(2), 553–557.
Mirseyed, P. S., Arjmand, S., Rahmandoust, M., Kheirabadi, S., & Anbarteh, R. (2024). Green synthesis of yeast cell wall-derived carbon quantum dots. Heliyon, 10(9), e29440.
Murru, C., Badía-Laíño, R., & Díaz-García, M. E. (2020). Synthesis and characterization of green carbon dots for scavenging radical oxygen species. Antioxidants, 9, 1147.
Nguyen, K. G., Baragau, I. A., Gromicova, R., Nicolaev, A., Thomson, S. A., Rennie, A., & Kellici, S. (2022). Investigating the effect of N-doping on carbon quantum dots. Scientific Reports, 12, 13806.
Pham, B., Tran, D. D., Nguyen, T. P. A., Nguyen, Q. K., Ung, T. D. T., Tran, N. B., & Pham, T. N. M. (2025). Detection of streptomycin using molecularly imprinted carbon dots. Spectrochimica Acta Part A, 343, 126622.
Prasannan, A., & Imae, T. (2013). One-pot synthesis of fluorescent carbon dots from orange waste peels. Industrial & Engineering Chemistry Research, 52(44), 15673–15678.
Praseetha, P. K., Litany, R. J., Alharbi, H. M., Khojah, A. A., Akash, S., Bourhia, M., & Shazly, G. A. (2024). Green synthesis of fluorescent carbon quantum dots from almond resin. Scientific Reports, 14(1), 24435.
Qi, H., Qiu, L., Zhang, X., Yi, T., Jing, J., Sami, R., & Rahman, M. M. (2023). N-doped carbon dots from citric acid and urea for metronidazole detection. RSC Advances, 13(4), 2663–2671.
Rao, L., Tang, Y., Lu, H., Yu, S., Ding, X., Xu, K., & Zhang, J. Z. (2018). Highly photoluminescent and stable N-doped carbon dots as nanoprobes for Hg2+ detection. Nanomaterials, 8(11), 900.
Rajurkar, N. S., & Hande, S. M. (2011). Estimation of phytochemical content and antioxidant activity of medicinal plants. Indian Journal of Pharmaceutical Sciences, 73(2), 146.
Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M., & Rice-Evans, C. (1999). Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine, 26(9-10), 1231–1237.
Redondo-Fernandez, G., Canga, J. C., Soldado, A., Encinar, J. R., & Costa-Fernandez, J. M. (2023). Functionalized heteroatom-doped carbon dots for biomedical applications: A review. Analytica Chimica Acta, 1284, 341874.
Roy, S., Ezati, P., & Rhim, J. W. (2021). Gelatin/carrageenan-based functional films with carbon dots. ACS Applied Polymer Materials, 3(12), 6437–6445.
Sekar, R., Basavegowda, N., Jena, S., Jayakodi, S., Elumalai, P., Chaitanyakumar, A., & Baek, K. H. (2022). Carbon dot-based photodynamic therapy for cancer. Pharmaceutics, 14(9), 1869.
Senel, B., Demir, N., Büyükköroglu, G., & Yıldız, M. (2019). Graphene quantum dots: Synthesis, characterization, cell viability, genotoxicity for biomedical applications. Saudi Pharmaceutical Journal, 27(6), 846-858.
Shabbir, H., Wojtaszek, K., Rutkowski, B., Csapó, E., Bednarski, M., Adamiec, A., & Wojnicki, M. (2022). Milk-derived carbon quantum dots. Molecules, 27(24), 8728.
Sun, Y. P., Zhou, B., Lin, Y., Wang, W., Fernando, K. S., Pathak, P., & Xie, S. Y. (2006). Quantum-sized carbon dots for photoluminescence. Journal of the American Chemical Society, 128(24), 7756–7757.
Wang, H., Sun, P., Cong, S., Wu, J., Gao, L., Wang, Y., ... & Zou, G. (2016). Nitrogen-doped carbon dots for “green” quantum dot solar cells. Nanoscale research letters, 11(1), 27.
Wang, S., Sun, W., Yang, D. S., & Yang, F. (2020). Soybean-derived photoluminescent carbon dots.Beilstein Journal of Nanotechnology, 11, 606–619.
Wechakorn, K., Khaopueak, P., Chobpattana, V., Sapermsap, N., Sangtawesin, S., & Sangtawesin, T. (2025). Sugar-derived carbon dot-functionalized glutathione for Hg2+ detection. ACS Omega, 10(5), 4496–4504.
Xu, X., Ray, R., Gu, Y., Ploehn, H. J., Gearheart, L., Raker, K., & Scrivens, W. A. (2004). Electrophoretic analysis of fluorescent carbon nanotube fragments. Journal of the American Chemical Society, 126(40), 12736–12737.
Yang, Z., Xu, M., Liu, Y., He, F., Gao, F., Su, Y., & Zhang, Y. (2014). Nitrogen-doped highly photoluminescent carbon dots from ammonium citrate. Nanoscale, 6(3), 1890–1895.
Yuan, F., Li, S., Fan, Z., Meng, X., Fan, L., & Yang, S. (2016). Shining carbon dots: synthesis and applications. Nano Today, 11(5), 565–586.
Zhang, M., Yuan, P., Zhou, N., Su, Y., Shao, M., & Chi, C. (2017). pH-sensitive N-doped carbon dots–heparin drug delivery system. RSC Advances, 7(15), 9347–9356.
Zhang, P., Wei, J. S., Chen, X. B., & Xiong, H. M. (2019). Heteroatom-doped carbon dots catalysts for oxygen reduction. Journal of Colloid and Interface Science, 537, 716–724.
Zhang, Q., Zhang, X., Bao, L., Wu, Y., Jiang, L., Zheng, Y., & Chen, Y. (2019). Green synthesis carbon quantum dots for mercury detection. Journal of Analytical Methods in Chemistry, 2019, 8183134.
Zhao, L., Zhang, M., Mujumdar, A. S., Adhikari, B., & Wang, H. (2022). Carbon dot/polyvinyl alcohol composite film for food preservation. ACS Applied Materials & Interfaces, 14(33), 37528–37539.
Zhu, Z., Li, Q., Li, P., Xun, X., Zheng, L., Ning, D., & Su, M. (2019). Surface charge controlled nucleoli selective staining with carbon dots. PLOS ONE, 14(5), e0216230.
Zulfajri, M., Gedda, G., Chang, C. J., Chang, Y. P., & Huang, G. G. (2019). Carbon dots as fluorescence sensor for Fe3+. ACS Omega, 4(13), 15382–15392.
Zulfajri, M., Sudewi, S., Rasool, A., Hsu, S. C., & Huang, G. G. (2023). Fluorescent ink and sensing of tartrazine using N-doped carbon dots. Waste and Biomass Valorization, 14(12), 3971–3986.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Faculty of Science, Burapha University

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Burapha Science Journal is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0) licence, unless otherwise stated. Please read our Policies page for more information




